|
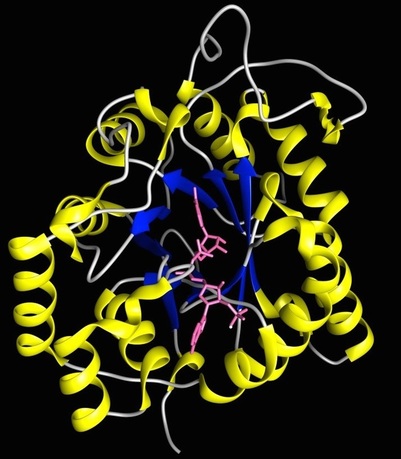
Enzyme Stereoselectivity and Stereospecificity: We would like to understand how the structure of a yeast reductase enzyme determines which mirror image of the alcohol (the left-handed or right-handed alcohol) is formed. Kayser, et.al., has found that the length of a region of the enzyme called Loop A (which stands for Substrate-Binding Loop A), plays an important role in determining whether the alcohol is left-handed or right-handed (we call this ability to preferentially form right-handed or left-handed molecules stereoselectivity and how well you make one or the other molecule is stereospecificity). However, we have found that this isn't the entire story. Changing the amino acid sequence of Loop A in a reductase called YDL124w altered the enzyme's ability to form the different-handed alcohols. We plan to explore the role of amino acid composition in more detail by mutating each amino acid in Loop A one at a time, and then determine how these individual changes affect the stereoselectivity of the mutant enzymes. This systematic approach will enable us to determine the role each individual amino acid in Loop A plays in determining enzyme stereoselectivity. Students will be exposed to the following techniques: Mutagenesis, DNA cloning, DNA expression and purification, polymerase chain reaction (i.e., PCR), protein expression and purification, restriction digests, bacterial and eukaryotic cell culture, and Gel electrophoresis.
|